PENNSYLVANIA, USA — A new COVID-19 drug for people who are immunocompromised is quickly making its way to Central Pennsylvania.
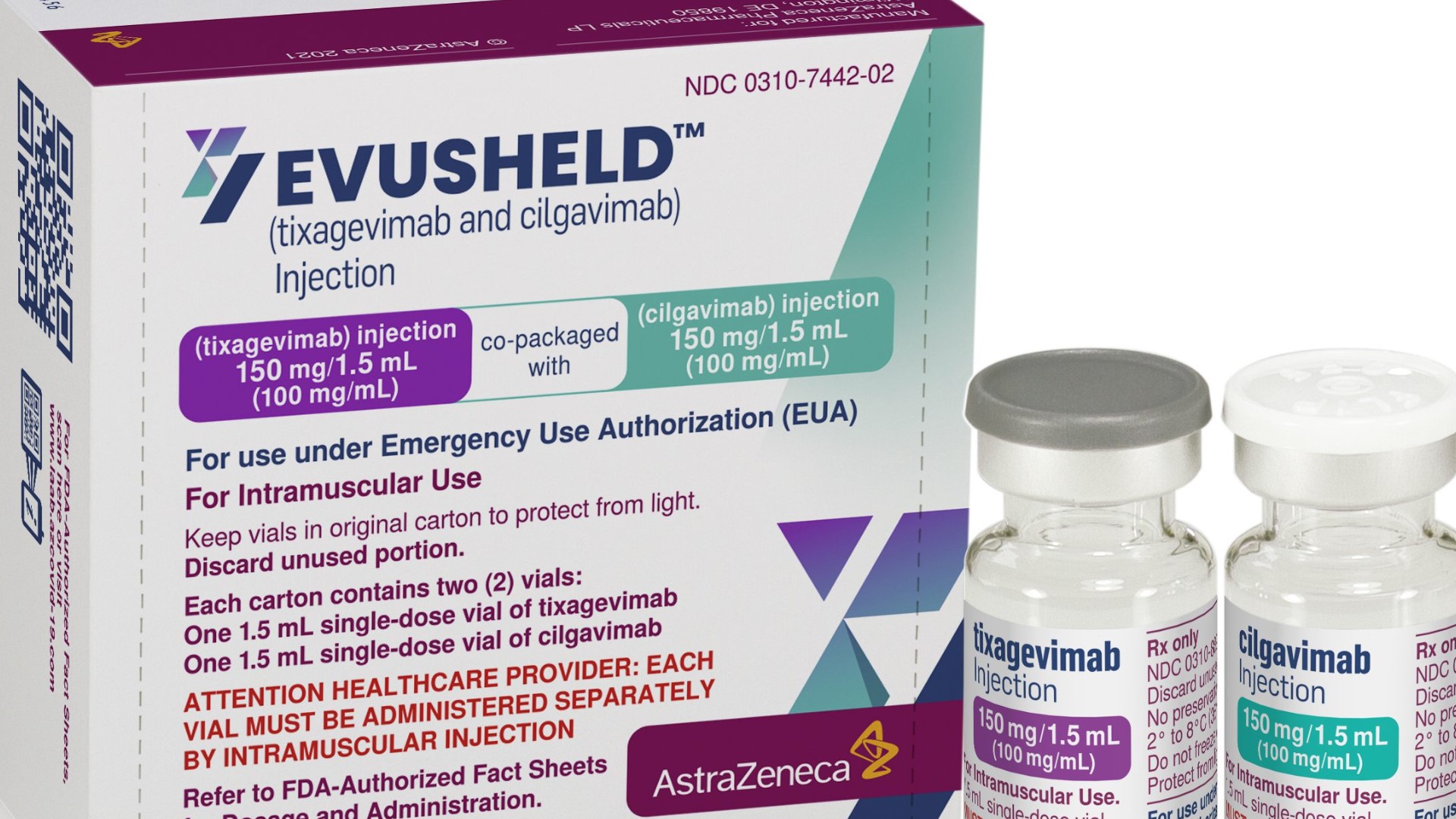
The FDA approved treatment called Evusheld is for people with weakened immune systems, such as organ transplant recipients, blood cancer patients or those who are allergic to the ingredients in the COVID-19 vaccines.
"Evusheld is a combination of two antibodies that's given in two muscular injections, one in each arm," said Dr. Erin McCreary, a University of Pittsburgh Medical Center( UPMC) Infectious Disease Specialist.
So far, UPMC, Penn State, Penn Medicine Lancaster and WellSpan Health have all received doses of the drug.
WellSpan Health did not disclose the quantity of the treatment the health system received but said that it is a small amount.
UPMC said it has received 456 doses for at least 800,000 patients that qualify. The hospital said the total is a fourth of the total supply that was allocated to Pennsylvania.
"So we can treat the sickest of the sick," said McCreary.
If you're rushing to get Evusheld, UPMC isn't offering the treatment on a first come, first serve basis, but a lottery.
"So what we know from our experience and our ethical practices is that first come first serve is not ethical because people that are more likely to access health care would come to you first. So you're disadvantaging patients that are less likely to access care," said McCreary.
In a statement UPMC said:
"We have categorized all EUA- eligible patients into three priority groups based on how likely the patients are to have a robust response to the vaccine or not. These groups were largely informed by our own research data, called the COVICS study, which looked at vaccine response in immunocompromised patients." - McCreary.
Instead of a lottery, WellSpan Health will utilize the prioritization criteria established by the National Institution of Health.
Hospitals said they are partnering with the Pennsylvania Department of Health and expect to receive more doses soon.